In aryl halides the halogen bearing carbon is part of.
What is a vinyl alkyl halide.
Halogens are treated the same way as alkyl groups.
Classified as primary secondary or tertiary according to the degree of substitution at the carbon to which the halogen is attached.
Finally halo is prefixed to it.
Structure and physical properties.
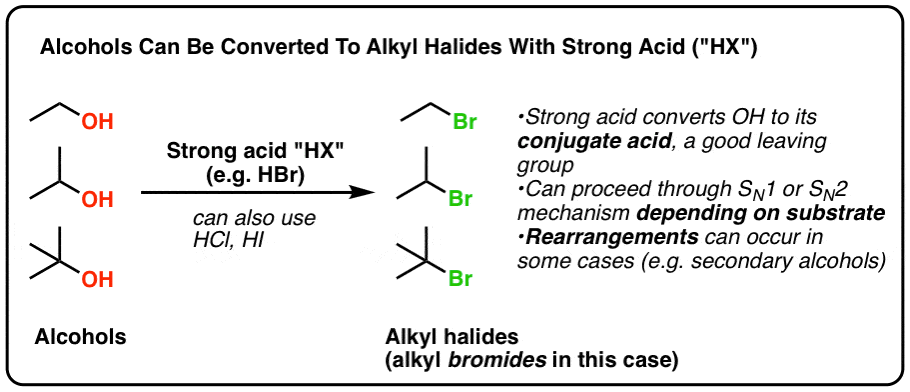
Alkyl halides also known as haloalkanes are compounds in which one or more hydrogen atoms in an alkane have been replaced by halogen atoms fluorine chlorine bromine or iodine.
There is an interaction between one of the lone pairs on the chlorine atom and the delocalized ring electrons and this strengthens the bond.
However alkyl halides may sometimes be confused with aryl halides.
Methyl iodide and ethyl chloride.
In this system a root word is chosen based on the number of carbon atoms present in the parent chain and then the primary suffix ane is added.
With the exception of iodine these halogens have electronegativities significantly greater than carbon.
Vinyl or more specifically pvc is also used in plastic pipes synthetic leather records hence the name vinyl record insulation and many other products.
For example if the halogen atom is attached to a carbon atom which is attached to a benzene ring cl ch 2 c 6 h 5 one would think it is an aryl halide but it is an alkyl halide because the halogen atom is attached to the carbon that is sp 3 hybridized.
Other articles where vinylic halide is discussed.
Other articles where tertiary alkyl halide is discussed.
They are subdivided into alkyl vinylic aryl and acyl halides.
Iupac nomenclature of alkyl halide or haloalkanes in substitutive system of iupac nomenclature the alkyl halides are named as haloalkanes.
Alkyl halides fall into different classes depending on how the ha.
In the generally accepted nomenclature of alkyl halides the name of the alkyl residue is followed by the name of the halide e g.
A vinyl halide is clearly a species with a formula h 2c c x h in which a halide is directly bound to an olefinic bond formally this is ethylene h 2c ch 2 with one of the hydrogens substituted by a heteroatom vinyl chloride h 2c chcl is an example.
Consequently this functional group is polarized so that the carbon is electrophilic and the halogen is.
Halogens are more electronegative than carbon.
In a primary alkyl halide the carbon that bears the halogen is directly bonded to one other carbon in a secondary alkyl halide to two and in a.
An aryl halide has general formula c 6h 5x in which an halide group x has substituted the aryl ring.
In alkyl halides all four bonds to the carbon that bears the halogen are single bonds.
In vinylic halides the carbon that bears the halogen is doubly bonded to another carbon.
The extra strength of the carbon halogen bond in aryl halides.
Start with an alkyl halide and.
The name of the halogen is followed by the name of the.